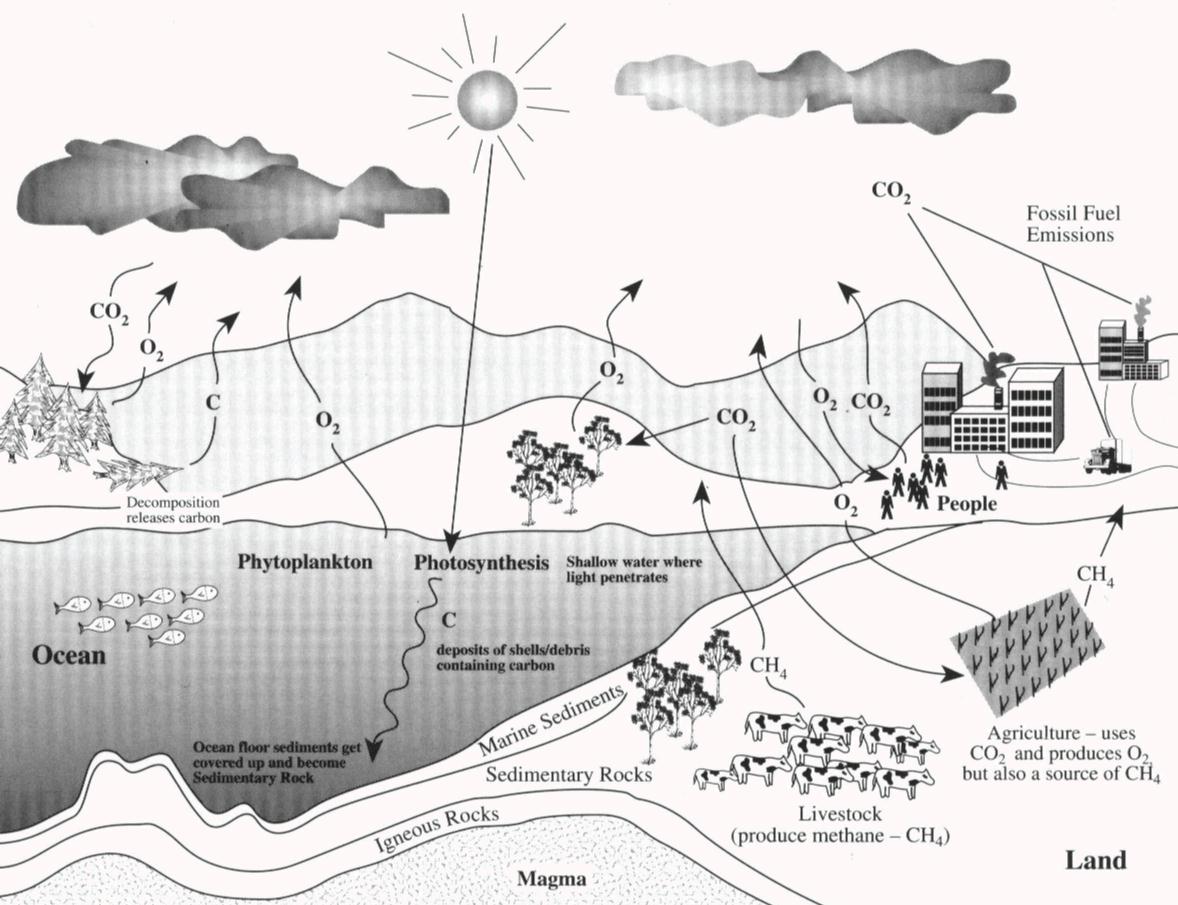
(f) the roles of respiration, combustion and photosynthesis in the maintenance of the levels of oxygen and carbon dioxide in the atmosphere.Unit 1: CHEMICAL SUBSTANCES, REACTIONS and ESSENTIAL RESOURCES.C6.3e describe the potential effects of increased levels of carbon dioxide and methane on the Earth’s climate and how these effects may be mitigated.C6.3 Interpreting and interacting with earth systems.C6.2e describe the potential effects of increased levels of carbon dioxide and methane on the Earth’s climate and how these effects may be mitigated.C6.2 Interpreting and interacting with earth systems.C1.3.4 describe how the effects of increased levels of carbon dioxide and methane may be mitigated, including consideration of scale, risk and environmental implications.C1.3 What is the evidence for climate change, why is it occurring?.8.26c Describe that these effects may be mitigated: consider scale, risk and environmental implications.8.26b Describe the potential effects on the climate of increased levels of carbon dioxide and methane generated by human activity, including burning fossil fuels and livestock farming.Students should be able to: describe actions to reduce emissions of carbon dioxide and methane.The carbon footprint can be reduced by reducing emissions of carbon dioxide and methane.The carbon footprint is the total amount of carbon dioxide and other greenhouse gases emitted over the full life cycle of a product, service or event.5.9.2.4 The carbon footprint and its reduction.5.9.2 Carbon dioxide and methane as greenhouse gases.Describe the potential effects of increased levels of carbon dioxide and methane on the Earth’s climate and how these effects may be mitigated, including consideration of scale, risk and environmental implications.4.4.1.5 Climate change: impacts and mitigation.Students should be able to recall a source for the nitrogen and a source for the hydrogen used in the Haber process.The raw materials for the Haber process are nitrogen and hydrogen.The Haber process is used to manufacture ammonia, which can be used to produce nitrogen-based fertilisers.4.10.4 The Haber process and the use of NPK fertilisers. 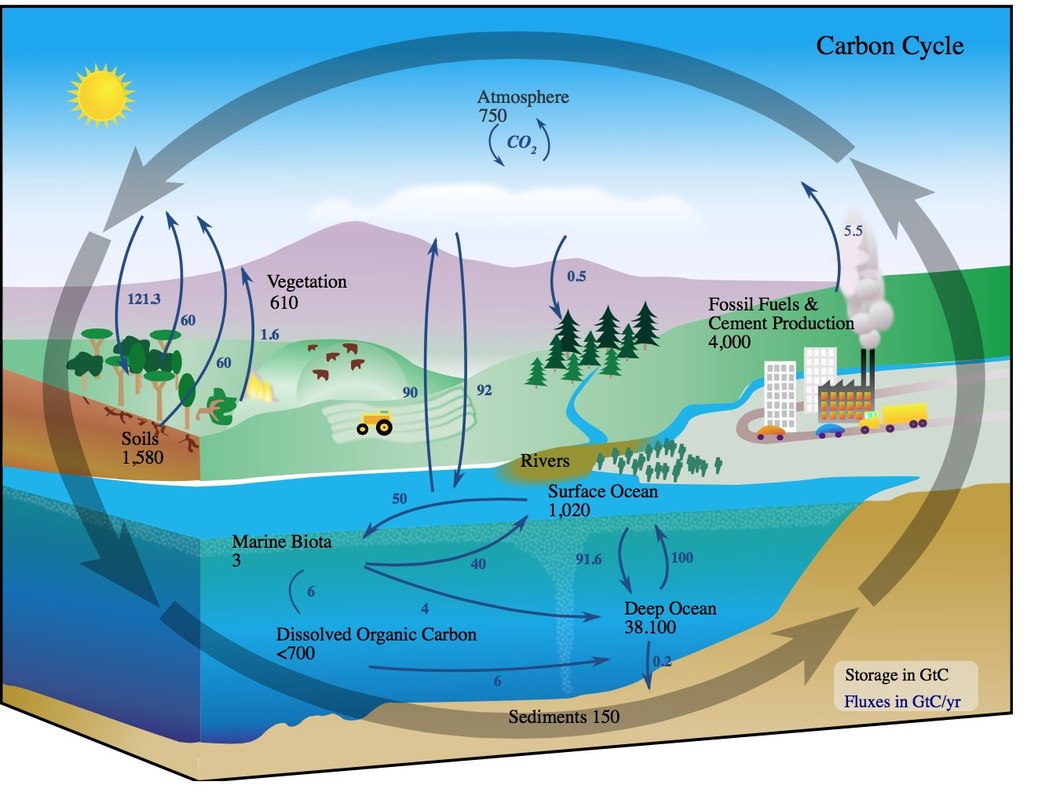
RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
